With more than three decades of experience in biotechnology and a record of reliably supplying medicines to patients,1 Amgen is known worldwide as a leader in research, development, and manufacturing of complex biologic therapies.
Shortages of prescription medicines are a growing problem around the world. Drug shortages can have a significant impact on patients and healthcare providers. As identified by the FDA, quality, manufacturing, and capacity issues are major reasons for many of these drug shortages.
We are deeply committed to ensuring doctors and patients can rely on the quality and availability of our treatments, including the biosimilars we plan to manufacture. The Amgen approach to providing a continuous supply of biologic therapies has four key elements:
- PREVENTION, which includes ensuring exemplary regulatory compliance, robust quality management processes, operational excellence, supply chain security, infrastructure investments, and business continuity planning
- TECHNOLOGY to enhance product purity and the robustness of the manufacturing process
- INVENTORY MANAGEMENT to ensure we have the right quantities of the product housed in diverse geographic locations
- REDUNDANT MANUFACTURING capacity in multiple geographic locations and back-up suppliers for raw materials
We've taken steps to understand potential vulnerabilities in processes and facilities and made significant investments to help ensure the quality and supply of Amgen biotech products.3
Reliability in Action
Drug shortages are a growing global concern. Beyond compliance with current good manufacturing practices and a robust quality management system, Amgen takes extra steps to help ensure a reliable supply of medicine to every patient, every time.

Mitigating Risk in Supply in an Era of Drug Shortages
As drug shortages become a growing global concern,1 leading manufacturers of biologics, such as Amgen, have identified key practices to help mitigate risk in supply to patients.
Causes of drug shortages can include manufacturing difficulties, product quality issues, manufacturing delays, unexpected increased product demand, product discontinuations and recalls, and raw material sourcing issues.1 Amgen has developed a multifaceted approach to help prevent drug shortages and employ risk management tactics that involve a continuous process of identifying, preventing, and responding to risks holistically.2
Beyond compliance with current good manufacturing practices (cGMP) and a robust quality management system, Amgen takes extra steps to help ensure a reliable supply to every patient, every time. As a first step, Amgen invests in inventory management at multiple points in the supply chain to mitigate potential risks of disruptions to supply (e.g., natural disasters). Additionally, Amgen maintains appropriate levels of raw materials by diversifying suppliers of sourced raw materials and storing high-risk raw materials in multiple geographical locations to safeguard their availability. By managing relationships with suppliers to ensure that raw materials requirements are understood and shared between both parties, Amgen is able to assure high-quality raw materials to minimize reprocessing and potential product manufacturing issues, and help ensure consistent product quality.2
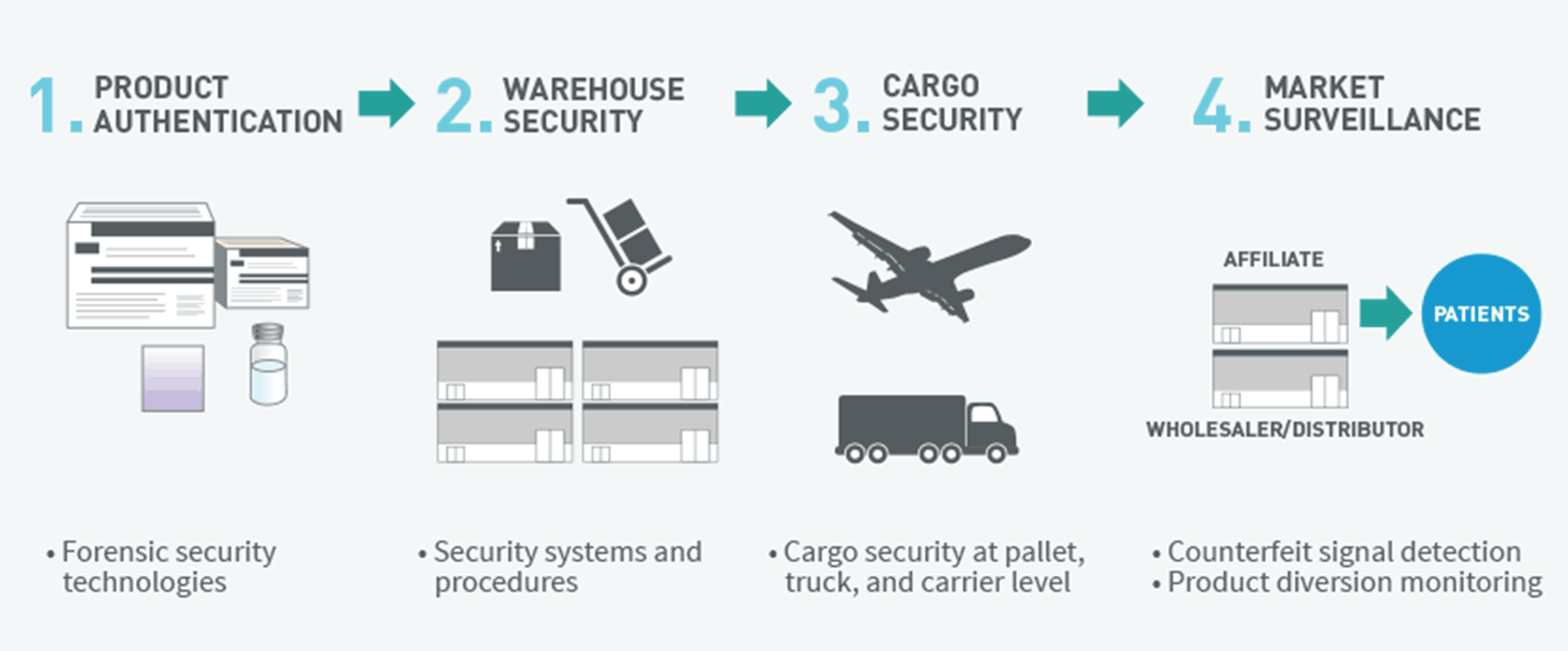
Maintaining diverse manufacturing capabilities requires significant investment in internal manufacturing and contract manufacturing capabilities.2 By maintaining eight manufacturing plants around the globe, Amgen is able to execute plans to implement back-up manufacturing capabilities (diversification), as well as enhance existing contract manufacturing capabilities.3 Amgen is also able to ensure the active management of robust and secure distribution networks, by establishing well-controlled temperature (“cold chain”) distribution channels to maintain quality of each product and utilizing multiple approaches to mitigate distribution-related security risks to supply. These include controlled shipping lanes using temperature-validated shipping containers to maintain chain of custody, anti-theft and anti-counterfeiting measures to mitigate the risk of diversion and adulteration, and risk management programs to systematically address risks as they develop.2
Amgen implements product security programs to help ensure reliable supply of its medicines to patients2
References
- Woodcock J, Wosinska M. Clinical Pharmacology & Therapeutics, Vol. 93, No. 2, February 2012.
- Mica A, Green L, American Society of Health-System Pharmacists, 2012, poster
- Data on file. Amgen.
