The dominant paradigm that has ruled drug discovery for decades is giving way to a new age of multispecific drugs that harness the power of biology.
By Raymond Deshaies
EDITOR’S NOTE
Beginning with the introduction of aspirin at the start of the 20th century, there have been three major waves of innovation in drug discovery. We are now at the start of a fourth wave that will radically alter our concept of how drugs can work, paving the way for new treatments aimed at thousands of disease targets now viewed as “undruggable.”
To appreciate the staggering potential of this emerging paradigm shift, let me briefly describe the first three waves and their impact on modern medicine. Wave 1, launched by aspirin, gave us medicines with well-defined chemical structures but unknown biology. They worked, but we didn’t know why they worked. In the 1970s, Wave 2 introduced rational drug design—specific compounds aimed at specific molecular targets that play a role in disease. Drug hunters finally knew not only what they were making but how it achieved its effects. The 1980s ushered in Wave 3, the biotechnology revolution. Recombinant DNA technology allowed us to go beyond chemical compounds and design medicines based on the proteins and other large molecules that govern biology.
While transformative in many ways, these innovations didn’t really change our basic idea of how drugs should work. In nearly every medicine approved dating back to aspirin, you have one molecule—your drug—and another molecule—your target. The drug binds to the target in a manner that changes the target’s normal activity. The drug itself does all the hard work of dialing the activity up or down, most often down. That’s drug design in a nutshell. Conceptually, it isn’t very complicated.
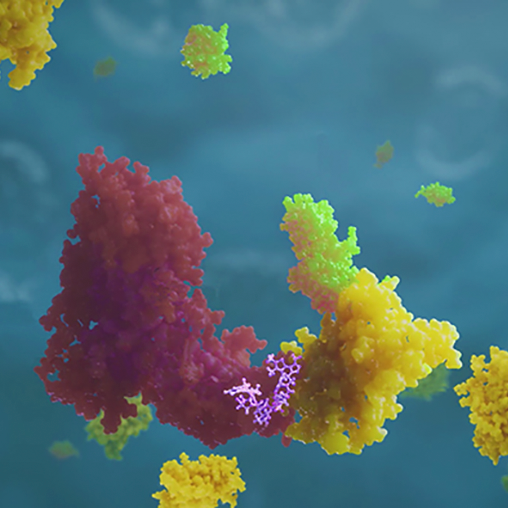
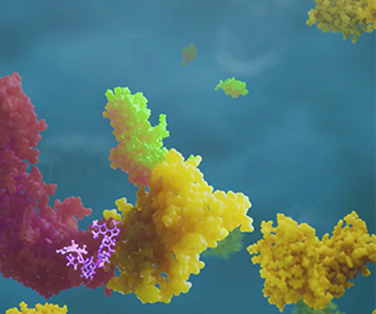
Future medicines could function very differently. In biopharma pipelines across the industry, we’re seeing more investigational multispecific drugs that can form connections with two or more proteins. They include some highly sophisticated structures that function as molecular matchmakers. Instead of engaging targets on their own, these agents mobilize biological mechanisms to do the heavy lifting. By inducing proximity between their targets and these mechanisms, multispecifics can harness the awesome power of biology to go well beyond what conventional drugs can accomplish. This isn’t an incremental improvement in drug design, it’s a sea change.
The molecular version of a recycling sticker
Nature’s playbook offers good examples of what induced proximity looks like in action, including some clever viruses that can commandeer the cell’s protein disposal system. When a cell wants to get rid of a protein, it uses specialized enzymes to tag the protein with ubiquitin—a molecular version of a recycling sticker. When ubiquitin-tagged proteins encounter a cellular machine called a proteasome, they get pulled inside and chopped into little pieces. Some viruses have learned how to thwart the cell’s antiviral proteins by making molecules that link these proteins to ubiquitin-tagging enzymes. Basically, they trick the cell into trashing its own defenses.
Drugs that can accomplish this same trick are called PROTAC® molecules (proteolysis targeting chimeras). A few medicines, including the successful cancer drug lenalidomide, were discovered to function in this manner only after they were approved and on the market. Now there are several companies investing in research into similar molecules, attracted by the large advantages these potential therapies might provide.
For starters, a conventional drug only works for as long as it’s binding to its target. The instant it loses its grip, the target goes back to causing disease. A PROTAC molecule doesn't need to remain in contact with its target because one encounter can trigger the target’s irreversible destruction. What’s more, just one of these molecules can take out multiple targets sequentially. Its effects are more durable because the target is gone until the cell can make more of it, which takes time. More importantly, these and other types of induced proximity agents could overcome a big limitation that narrows the range of targets current drugs can hit.
Just as a key needs a matching lock to work, conventional drugs need targets with binding pockets that are essential to the target’s activity. Proteins lacking functional pockets that drugs can nestle into are deemed “undruggable”—and about 85 percent of human proteins fall into this category. However, since the effectiveness of PROTAC molecules come from the enzymes they bring to the party, their usefulness isn’t restricted to the 15 percent of proteins with functional pockets. Instead of fitting like a key in a lock, they can use any binding site that allows their companion enzymes to do their job.
Limitless possibilities
PROTAC molecules are just the tip of the iceberg in terms of the matchmaker molecules we could design. In biology, countless mechanisms work through induced proximity, which means there are countless ways for drug hunters to try to treat disease by exploiting this approach.
- Already, we’re seeing early descriptions of “LYTAC” molecules, which can link targets in the blood to the receptors that cells use to capture and recycle unwanted proteins.
- The target doesn’t need to be a protein. Cells have specialized machinery to eliminate unwanted RNA, the nucleic acid that translates DNA into proteins. “RIBOTAC” molecules could eliminate specific RNA sequences linked to disease proteins.
- We could also design “AUTAC” molecules that leverage a process called autophagy (literally, “self-eating”) that cells use to gobble up unwanted debris, like clumps of sticky proteins and damaged organelles.
The potential for generating new combinations and new biology is endless. We’re only limited by our technical capabilities, vision, and creativity.
At Amgen, we addressed a key technical need in 2019 by acquiring Nuevolution, now Amgen Research Copenhagen. The scientists there are world leaders in building DNA-encoded libraries. This technology uses DNA tags to barcode individual chemical compounds in huge mixtures of billions of different compounds. The Copenhagen team has built libraries that collectively hold 40 trillion unique compounds. That massive collection will help us to find those one-in-a-billion compounds needed to reach the high-hanging fruit of potential drug targets using multispecific molecules.
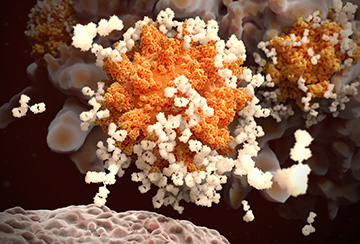
This isn’t the first time Amgen has sought to build a highly promising platform technology. In 2012, we acquired Micromet, which pioneered a new type of therapy known as bispecific T cell engagers, or BiTE® molecules. These molecules work by inducing proximity at the cellular level, bringing T cells together with tumor cells so the former can recognize and destroy the latter. Eight years after that acquisition, we have one approved BiTE therapy, additional BiTE molecules targeting seven types of cancer now in clinical trials, and third-, fourth-, and fifth-generation BiTE structures at the design stage. In the decade ahead, we hope to make similar progress with our small molecule induced proximity platform.
The urgency of this effort is underscored by many discoveries emerging from human genetics and allied data. These technologies are revealing that numerous genes and proteins that lead to disease are linked to targets that can’t be drugged by any conventional medicine. Multispecific molecules have the potential to expand the universe of druggable proteins from just 15 percent to nearly 100 percent. This new wave of innovation makes it possible to imagine a day when many more diseases can be treated or even cured.
Amgen Forward-Looking Statements
This communication contains forward-looking statements that are based on the current expectations and beliefs of Amgen. All statements, other than statements of historical fact, are statements that could be deemed forward-looking statements, including any statements on the outcome, benefits and synergies of collaborations, or potential collaborations, with any other company, including Adaptive Biotechnologies (including statements regarding such collaboration’s ability to discover and develop fully-human neutralizing antibodies targeting SARS-CoV-2 to potentially prevent or treat COVID-19), BeiGene, Ltd., or the Otezla® (apremilast) acquisition, including anticipated Otezla sales growth and the timing of non-GAAP EPS accretion, as well as estimates of revenues, operating margins, capital expenditures, cash, other financial metrics, expected legal, arbitration, political, regulatory or clinical results or practices, customer and prescriber patterns or practices, reimbursement activities and outcomes, effects of pandemics or other widespread health problems such as the ongoing COVID-19 pandemic on our business, and other such estimates and results. Forward-looking statements involve significant risks and uncertainties, including those discussed below and more fully described in the Securities and Exchange Commission reports filed by Amgen, including our most recent annual report on Form 10-K and any subsequent periodic reports on Form 10-Q and current reports on Form 8-K. Unless otherwise noted, Amgen is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise.
No forward-looking statement can be guaranteed and actual results may differ materially from those we project. Our results may be affected by our ability to successfully market both new and existing products domestically and internationally, clinical and regulatory developments involving current and future products, sales growth of recently launched products, competition from other products including biosimilars, difficulties or delays in manufacturing our products and global economic conditions. In addition, sales of our products are affected by pricing pressure, political and public scrutiny and reimbursement policies imposed by third-party payers, including governments, private insurance plans and managed care providers and may be affected by regulatory, clinical and guideline developments and domestic and international trends toward managed care and healthcare cost containment. Furthermore, our research, testing, pricing, marketing and other operations are subject to extensive regulation by domestic and foreign government regulatory authorities. We or others could identify safety, side effects or manufacturing problems with our products, including our devices, after they are on the market. Our business may be impacted by government investigations, litigation and product liability claims. In addition, our business may be impacted by the adoption of new tax legislation or exposure to additional tax liabilities. If we fail to meet the compliance obligations in the corporate integrity agreement between us and the U.S. government, we could become subject to significant sanctions. Further, while we routinely obtain patents for our products and technology, the protection offered by our patents and patent applications may be challenged, invalidated or circumvented by our competitors, or we may fail to prevail in present and future intellectual property litigation. We perform a substantial amount of our commercial manufacturing activities at a few key facilities, including in Puerto Rico, and also depend on third parties for a portion of our manufacturing activities, and limits on supply may constrain sales of certain of our current products and product candidate development. An outbreak of disease or similar public health threat, such as COVID-19, and the public and governmental effort to mitigate against the spread of such disease, could have a significant adverse effect on the supply of materials for our manufacturing activities, the distribution of our products, the commercialization of our product candidates, and our clinical trial operations, and any such events may have a material adverse effect on our product sales, business and results of operations. We rely on collaborations with third parties for the development of some of our product candidates and for the commercialization and sales of some of our commercial products. In addition, we compete with other companies with respect to many of our marketed products as well as for the discovery and development of new products. Discovery or identification of new product candidates or development of new indications for existing products cannot be guaranteed and movement from concept to product is uncertain; consequently, there can be no guarantee that any particular product candidate or development of a new indication for an existing product will be successful and become a commercial product. Further, some raw materials, medical devices and component parts for our products are supplied by sole third-party suppliers. Certain of our distributors, customers and payers have substantial purchasing leverage in their dealings with us. The discovery of significant problems with a product similar to one of our products that implicate an entire class of products could have a material adverse effect on sales of the affected products and on our business and results of operations. Our efforts to collaborate with or acquire other companies, products or technology, and to integrate the operations of companies or to support the products or technology we have acquired, may not be successful. A breakdown, cyberattack or information security breach could compromise the confidentiality, integrity and availability of our systems and our data. Our stock price is volatile and may be affected by a number of events. Our business performance could affect or limit the ability of our Board of Directors to declare a dividend or our ability to pay a dividend or repurchase our common stock. We may not be able to access the capital and credit markets on terms that are favorable to us, or at all.